Ocean Chemical Processes
Why is the sea salty? Sea water contains about 35 grams per kilogram of dissolved salt. The most obvious source for the salt is river water, which can easily be observed weathering rocks (from which the water derives minerals), carrying sediment , and flowing continually into the ocean. * Because the water added to the ocean evaporates but the dissolved salts do not, it seems reasonable to suggest that river water brings salt to the ocean.
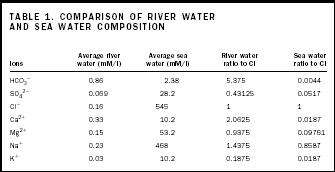
But a closer look shows that the process must be more complicated. Table 1 compares the major substances dissolved in river water and ocean water. If sea water is simply concentrated river water, these elements should be present in the same ratios in both types of water. For both water types, the Cl/Cl ratio is 1 because that is the chosen standard of comparison. Notice that the ratio patterns for most components for the two water types are quite different. This pattern means that simple evaporation of water cannot change river water into sea water.
Addition–Removal Processes and Considerations
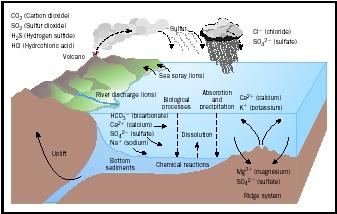
The composition of sea water is controlled by many different processes, all acting at the same time, and adding and removing substances at different rates (see figure). The sum of all the processes, a kinetic (changing) balance, determines sea-water chemistry (see Table 2 on page 136).
When sea water dries up completely, it leaves behind a salt deposit called an evaporite . Evaporites of greatly different ages on Earth all are similar,
| TABLE 1. COMPARISON OF RIVER WATER | ||||
| AND SEA WATER COMPOSITION Average river | Average sea | River water | Sea water | |
| Ions | water (mM/l) | water (mM/l) | ratio to Cl | ratio to Cl |
| Hco 3 − | 0.86 | 2.38 | 5.375 | 0.0044 |
| So 4 − | 0.069 | 28.2 | 0.43125 | 0.0517 |
| Cl − | 0.16 | 545 | 1 | 1 |
| Ca 2+ | 0.33 | 10.2 | 2.0625 | 0.0187 |
| Mg 2+ | 0.15 | 53.2 | 0.9375 | 0.09761 |
| Na + | 0.23 | 468 | 1.4375 | 0.8587 |
| K + | 0.03 | 10.2 | 0.1875 | 0.0187 |
so scientists reached the conclusion that sea water must have had roughly the same chemistry over hundreds of millions of years.
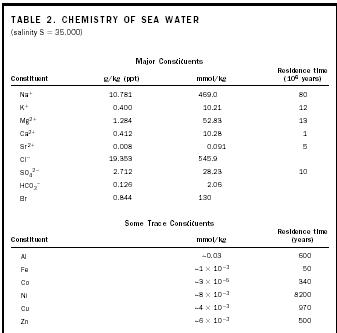
If this is true, then all the processes affecting sea-water chemistry must be at steady state—that is, operating so that the input of salt equals the output. For a steady-state ocean, it is possible to find out how long a particular element stays in the ocean (i.e., its residence time) before it is removed. The ocean is at steady state for a particular element if that element is added and removed at the same rate.
For example, Na (sodium) is added to the ocean at the rate of about 7.9 × 10 12 moles per year (3.4 × 10 5 tons per year) The whole ocean contains 6.4 × 10 20 moles (2.8 × 10 13 tons) of Na, and the total amount of Na present divided by the rate of addition gives 80 × 10 6 years, the residence time of Na. In other words, a sodium atom entering the ocean in river water will stay in the ocean for 80 × 10 6 years before it is removed from sea water.
Residence Time.
The concept of residence time is informative in several ways. Elements with long residence times in the ocean tend to be very soluble in sea water and to be evenly mixed throughout the ocean. Thus, Na, Cl, and other elements (Table 2) have long residence times and are known as conservative elements, occurring in the same ratio to one another throughout the ocean regardless of the total salinity.
Elements with short residence times (such as iron and aluminum) are relatively reactive, or insoluble in sea water; they are easily removed and are unevenly distributed throughout the ocean. This makes sense in comparison with the mixing time for the whole ocean, which is about 1,000 years. An element that remains dissolved for millions of years will have been mixed through the ocean many times over, and hence should be evenly distributed. In contrast, an element with a residence time of only 100 years will not be able to make it around once without being removed.
Estuaries.
Rivers add huge amounts of dissolved materials to the ocean each year as well as many tons of soil and rock particles. At the boundary between the land and the sea are estuaries, or bodies of water that are
| TABLE 2. CHEMISTRY OF SEA WATER | ||||||
| (salinity S = 35.000) | ||||||
| Major Constituents | Residence time | |||||
| Constituent | g/kg (ppt) | mmol/kg | (10 6 years) | |||
| Na + | 10.781 | 469.0 | 80 | |||
| K + | 0.400 | 10.21 | 12 | |||
| Mg 2+ | 1.284 | 52.83 | 13 | |||
| Ca 2+ | 0.412 | 10.28 | 1 | |||
| Sr 2+ | 0.008 | 0.091 | 5 | |||
| Cl − | 19.353 | 545.9 | ||||
| SO 4 2− | 2.712 | 28.23 | 10 | |||
| HCO 3 − | 0.126 | 2.06 | ||||
| Br | 0.844 | 130 | ||||
| Some Trace Constituents | Residence time | |||||
| Constituent | mmol/kg | (years) | ||||
| Al | ∼0.03 | 600 | ||||
| Fe | ∼1 × 10 −3 | 50 | ||||
| Co | ∼3 × 10 −5 | 340 | ||||
| Ni | ∼8 × 10 −3 | 8200 | ||||
| Cu | ∼4 × 10 −3 | 970 | ||||
| Zn | ∼6 × 10 −3 | 500 | ||||
chemical and physical transitions between rivers and oceans. As salinity and pH increase seaward, some dissolved substances, such as Fe (iron), may precipitate to form solids and then remove other dissolved elements, such as Mn (manganese), onto their surfaces. Other elements that arrive in the estuary adsorbed (bound) to river particle surfaces may be desorbed (unbound) by the influence of the higher salt content they encounter in the ocean. The dissolved substances enter the ocean, but as much as 90 percent of river-borne particles are trapped in the estuary and on the continental shelf .
Aerosols.
Particles carried through the air are known as aerosols. They come from a variety of sources. Natural aerosols include sea-spray residues, windblown soil particles, volcanic particles, smoke from forest fires, and particles condensed from natural gases. Anthropogenic (human-derived) aerosols, often considered pollutants, include direct emissions such as from smokestacks and particles from conversion of anthropogenic gases.
Because most of the river-derived sediment load is trapped in estuaries and on the continental shelf, a large fraction of particles reaching the ocean from land consists of aerosols. The global mineral dust source is 100 × 10 12 grams per year to 800 × 10 12 grams per year [(3.5 × 10 12 ounces per year to 28.2 × 10 12 ounces per year)], compared to a river discharge of 15.5 × 10 15 grams per year [(0.6 × 10 15 ounces per year)]. * Because most of the continental land mass is in the Northern Hemisphere, most of the natural and human-derived aerosols also are generated in the Northern Hemisphere. The amounts in air vary over time and tend to be concentrated in latitude zones.
Aerosols can be delivered to the ocean as dry fallout or as wet fallout if they are entrained into falling rain; in fact, most of the chloride (Cl − ) and sulfate (SO 4 2− ) in sea water is believed to have come from volcanic gases that were dissolved in rain and delivered to the ocean over Earth's history.
Hydrothermal Processes.
Sea water continually reacts with its "container"—the basalt rocks that underlie the ocean. The most intense of these reactions occur in hot springs along mid-ocean ridges. * The entire volume of ocean water gradually circulates into the ocean floor, reacts with hot basalt, and returns greatly changed in chemical composition.
In this transit, sea water loses all its O 2 and all its Mg; further, all its SO 4 2− is stripped of oxygen. At the same time, the water gains Ca, Si, Fe, and Mn. When the hot, altered hydrothermal fluids mix with cold, normal sea water, Fe, Mn, and Si precipitate out in huge plumes that look like smoke: hence the names " black smoker " and " white smoker ". * The solids formed in this precipitation remove trace metals (such as Cr, V, Mo, U) from solution. (Trace elements occur at concentrations of less than 1 part per million (ppm), or 1 milligram per kilogram of water.)
It is difficult to estimate how fast ocean water circulates through the mid-ocean ridge axis areas; a reasonable estimate of once every 10 million years suggests the importance of these reactions in establishing the composition of the ocean.
Biological Processes.
Life processes have an important effect on ocean chemistry. Certain surface-dwelling phytoplankton (e.g., coccolithophores and diatoms) remove calcium or silicon from sea water to make calcium carbonate shells or opalline silica shells, respectively. These hard particles eventually fall toward the ocean floor, where they are buried in the sediment and thus removed from sea water for many millions of years. The carbon removed by these same organisms to make their soft tissue becomes food for both higher organisms and for bacteria.
The carbon plus the Sun's energy make up the basic fuel for the biogeochemical cycling of material in the ocean. It may be difficult to imagine how an organism as small as a coccolithophore can affect the Ca content of the whole ocean until one considers the enormous numbers of these phytoplankton that occur in the surface ocean. *
In addition to the biological debris that falls through the ocean as discrete particles and fluffy bits of marine snow, many inorganic particles from soils are caught up in the fecal pellets and other biogenic materials. * All these particles have surfaces that may adsorb dissolved material from sea water and cause them to be buried in ocean floor sediments, thus removed from the ocean.
Evaporation.
Evaporation of isolated bodies of sea water accounts for removal of large amounts of salt from the ocean. Salt deposits occur through the geologic record as layers of gypsum, rock salt, sylvite, and other minerals. These minerals represent dissolved salts that have been removed from sea water by evaporation and are mined for table salt and other uses. *
Pore-Water Interactions.
Interaction with pore water can affect the chemistry of the ocean. Pore water is sea water that has been trapped between sediment grains. The chemistry of the pore water is susceptible to change by biological processes. For example, bacteria in the sediment consume organic tissue, at the same time using up much or even all the oxygen in the pore water.
In the absence of oxygen (O 2 ), various members of the bacterial population continue to metabolize their food using other energy sources; common examples are reduction of SO 4 2− to S − , reduction of Fe(OH) 3 to Fe 2+ , and reduction of Mn 4+ to Mn 2+ . Changed forms of the elements (such as Mn 2+ ) in some cases become soluble and diffuse out of the sediment into the overlying sea water; O 2 diffuses from sea water into the sediment. These processes are slow but continuous, and in the long term affect the chemistry of sea water.
Conclusion.
Many questions concerning the chemistry of sea water remain unanswered. In recent years, work on the composition of tiny sea-water bubbles included in salt crystals from ancient evaporites suggests that the composition of sea water may have changed over the past 700 million years. The changes amount to about a factor of two for several of the major elements; these observations will lead to much future research on the processes that establish ocean chemistry and their changes over time.
SEE ALSO Algal Blooms in the Ocean ; Brines, Natural ; Carbon Dioxide in the Ocean and Atmosphere ; Mid-Ocean Ridges ; Mineral Resources from the Ocean ; Ocean Biogeochemistry ; Plankton ; Sea Water, Freezing of ; Sea Water, Physics and Chemistry of ; Tracers of Ocean-Water Masses .
Martha R. Scott
Bibliography
Broecker, Wallace. S., and T. H. Peng. Tracers in the Sea. Palisades, NY: LDGO Press, 1982.
Chester, Roy Marine Geochemistry, 2nd ed. London, U.K.: Blackwell Publishers, 2000.
Dasch, E. Julius, ed. Encyclopedia of Earth Sciences. New York: Macmillan Reference USA, 1996.
Libes, Susan. An Introduction to Marine Biogeochemistry. New York: John Wiley & Sons, 1991.
Pilson, Michael E. Q. An Introduction to the Chemistry of the Sea. Upper Saddle River, NJ: Prentice Hall, 1998.
* See "Algal Blooms in the Ocean" for a photograph of a coccolithophore bloom.
* See "Hot Springs on the Ocean Floor" for a photograph of a black smoker.
* See "Mid-Ocean Ridges" for a composite image of the mid-ocean ridge system.
* See "Mineral Resources from the Ocean" for a photograph of mined stacks of sea salt.
* See "Ocean Biogeochemistry" for a photograph of marine snow.
* See "Ocean-Floor Sediments" for a photograph of dust blowing into the Red Sea.
* See "Oceanography from Space" for a photograph of a river in Borneo adding water and sediment to the coastal ocean.